Since his second term started, President Trump has announced, negotiated, or floated a flurry of initiatives aimed at taming the excesses of the pharmaceutical industry.
No surprise. About 60% of American adults are “worried about being able to afford prescription drug costs for themselves or their families,” a recent KFF nationwide poll showed. More than 80% consider the price of prescription drugs “unreasonable,” and most support increased regulation to lower costs. Americans pay about three times as much as people in other countries for the same prescription drugs.
Last July, Trump sent letters to 17 drugmakers, demanding they voluntarily lower drug prices. Then the president said he’d negotiated with more than a dozen pharmaceutical executives one by one at the White House. In December, he announced that he had compelled them to agree to “most favored nation” pricing on Medicaid, the government coverage for low-income Americans.
Then came the unveiling of TrumpRx, a site where cash-paying patients could find discounted medicines, and a promise to speed biosimilar products — generic versions of certain high-priced specialty drugs — by cutting through FDA red tape.
The scope of these promises remains uncertain. But it’s certainly less than what the announcement promised, partly because many details of the negotiations, even which drugs are covered, are hazy.
White House spokesperson Kush Desai did not answer queries about TrumpRx.
Medicaid already buys drugs at deep discounts. And other patients may well have better options through commercial drug discount programs, which offer far more products, or through their insurance and associated drug company copayment cards.
So, the share of Americans likely to benefit from these options remains slim, even if some people do come out ahead.
“If it makes a difference to any patient, it’s a win,” said Mark Cuban, a billionaire investor on his own mission to bring down drug prices. He pointed to discounted pricing on TrumpRx for branded fertility drugs and GLP-1 weight loss drugs for people without insurance or whose plans don’t include coverage. Cuban launched the Mark Cuban Cost Plus Drug Co., known as Cost Plus Drugs, in 2022 to sell drugs cheaply by eliminating middlemen — buying from factories and selling directly to consumers. Most of the drugs he sells are generics.
Aaron Kesselheim, a professor of medicine at Harvard Medical School whose research focuses on drug prices, said the Trump announcements are “one-off agreements made for publicity purposes. They don’t change anything about the way drugs are priced.”
He added: “The agreements are opaque and unenforceable.”
It was unclear, for example, which drugs would be sold at “most favored nation” prices or how exactly that was defined. But, clearly, not all were.
Doing the math
46brooklyn, a consulting firm and data project that tracks brand-name drug prices, found that close to 1,000 brand drugs went up in price in January 2026. What’s more, 2025 had the highest number of list price increases ever. “This is not a material change, it’s business as usual,” said Antonio Ciaccia, the company’s co-founder.
In the first week of 2026, Pfizer raised the list prices of 71 drugs by an average of 5% and lowered the price of only one, by 9.8%, the data project found.
The biggest win for patients has likely been the Trump administration’s quiet continuation of a Biden administration program: Medicare drug price negotiation for expensive drugs. The negotiated discounts on the initial 10 drugs — from blood thinners to insulins to medicines for inflammatory disorders — went into effect Jan. 1. With reductions in price of well over 50% on some products, the estimated $6 billion in annual savings allowed the program to cap Medicare patients’ out-of-pocket spending on Part D prescription drugs at $2,000 for 2025 and beyond.
An additional 15 high-priced drugs — including popular weight loss and cancer drugs — were subject to negotiation in 2025, with discounted Medicare prices taking effect next year. And 15 more high-priced drugs are set for negotiation this year. All told, the 40 negotiated drug prices are expected to save Medicare well over $20 billion a year.
Even as these discounts take effect, drug industry lobbyists have been working to limit the impact, with some success. For example, the One Big Beautiful Bill Act exempts drugs for rare diseases from negotiations.
Still, “this is historic because it’s the first time the United States has negotiated prices, like every other developed country,” Kesselheim said. “And guess what? Innovation didn’t stop.”
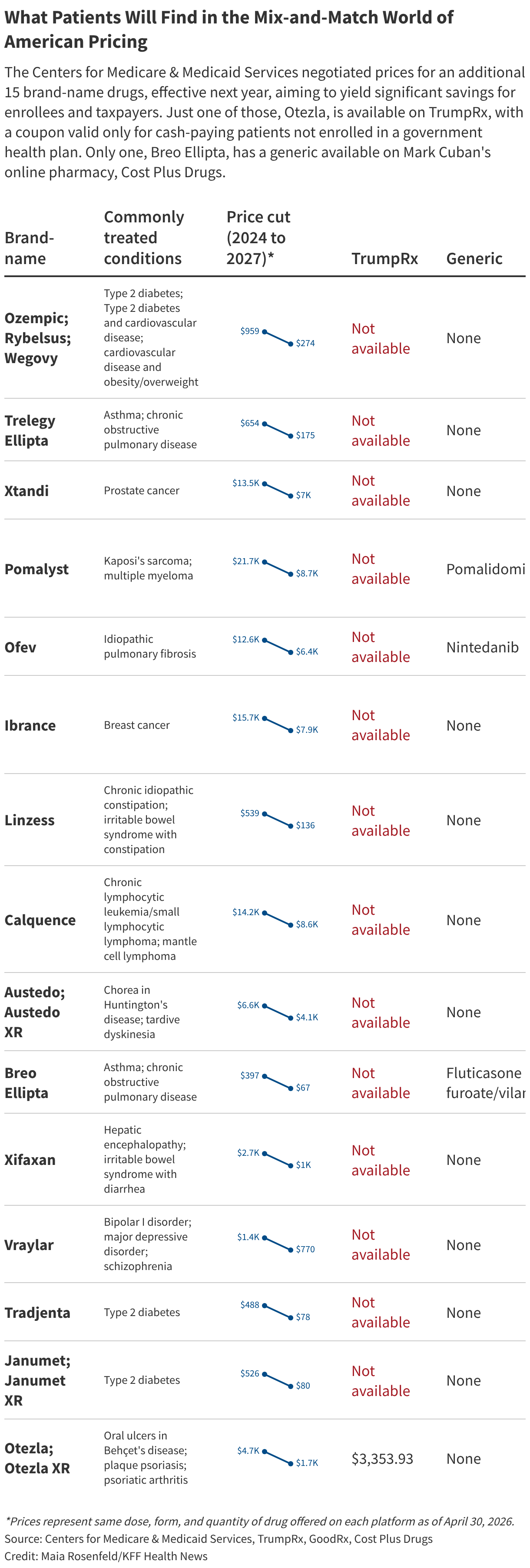
Of course, these discounts benefit only Medicare enrollees. The newer Trump administration initiatives help some other patients, but they are limited and require knowledge of how to access the discounts.
Trump’s one-on-ones
The president’s televised appearances with the heads of major drug companies resulted in deals, but few, if any, will mean much to patients. For example, after Trump met with Albert Bourla, CEO of Pfizer, the company announced discounts on 30-plus drugs. Bourla called the deal “a win for American patients, a win for American leadership, and a win for Pfizer.”
The discounts are offered via TrumpRx, which, in turn, offered coupons co-branded on GoodRx.com, which already offers discount coupons for many hundreds of medicines.
Pfizer made hay of the deal, announcing it was part of Pfizer’s broader, landmark most favored nation, or MFN, agreement with the U.S. government, enabling patients to pay lower prices for their prescription medicines “while strengthening America’s role as the global leader in biopharmaceutical innovation.”
Pfizer spokesperson Steven Danehy cited a press release from September, noting that the TrumpRx.gov site offers patients savings that “range as high as 85%.”
Most of the list features brand-name drugs, competing with far cheaper generic versions from other manufacturers, such as the cholesterol-lowering drug Colestid, which TrumpRx lists for “50% off” at $127.91. Generic versions cost about $17 on the Cost Plus site.
This means the branded companies aren’t making a sacrifice by offering them at lower costs as reflected on Trump’s portal, said Sean Tu, a patent law expert at the University of Alabama. “That’s a sale they would not have made if not for TrumpRx.”
Others are very old drugs, such as Cortef, or hydrocortisone, whose 5-milligram branded Pfizer version is listed at $45 on TrumpRx, half its list price of $91.80. It sells for far less on Cuban’s Cost Plus site. Still others, such as the $607.20 HIV treatment Viracept, are useful only in combination with other drugs that are not discounted.
Last week, TrumpRx added Amgen’s Humira, for years the world’s best-selling drug, at $950 a dose, down from a list price of nearly $7,000. But Humira lost its patent protection in 2023, and biosimilars — essentially generic equivalents — have since come to market. More to the point, two of those biosimilars are listed on TrumpRx for as little as $207.60 a dose.
Since most of the TrumpRx products are available only to customers without insurance who pay cash, the arthritis drug Xeljanz’s drop from $2,277 to $1,518 a month would still leave it unaffordable.
A few notable deals
The much-touted TrumpRx site, launched Feb. 6, consists largely of Pfizer’s 30 drugs (30 of roughly 85) with a smattering of discounts likely to generate headlines.
These include three fertility drugs from EMD Serono, a subsidiary of the pharmaceutical giant Merck KGaA, the most expensive of which, Gonal-F, has a list price of $966 but is only $168 per IVF cycle using a TrumpRx coupon.
They will save women thousands of dollars — although the overall cost of fertility treatment will continue to put them beyond the reach of many, since drugs represent only a portion of the payment.
The TrumpRx discounts could reduce the $15,000-to-$25,000 cost of a single fertility treatment cycle — women typically need two or three cycles to become pregnant — by about 10%, said Sean Tipton, spokesperson for the American Society for Reproductive Medicine. In some European countries, each cycle costs about $3,000.
In exchange for lowering those prices, EMD Serono got tariffs lifted on its mostly overseas-produced medications. It also won the right to a sped-up FDA approval process for a fertility drug it’s been marketing heavily in Europe.
Another newsworthy offering on the site resulted from a deal with Novo Nordisk for Wegovy, its GLP-1 drug for weight loss and diabetes, with the price reduced to as little as $199 a month for the pen. (Many insurers cover such drugs only for diabetes, leaving those who are interested in losing weight paying out-of-pocket. Zepbound, Wegovy’s Lilly & Co. competitor, is also on the list, at $299.)
Pressure has been building on Novo and Lilly to lower the U.S. price of their GLP-1 drugs. The compounds have lost patent protection in India, and pressure from customers buying overseas will likely increase when generic Wegovy goes on sale in Canada, for as low as $73 a month, possibly this year.
In the United States, meanwhile, dozens of patents should keep Wegovy generics off the market until 2039, said professor Robin Feldman, a patent expert at the University of California Law-San Francisco. A recent report from the research group I-Mak delved into several ways patent manipulation keeps generics off the U.S. market long after they are available in European countries and Canada.
And while the Trump administration has vowed to approve biosimilars more rapidly to ensure more competition and lower prices, that may not have much impact. The big hurdle in getting generics and biosimilars to market is often not FDA approval, but the time it takes to override the thickets of patents that U.S. law allows manufacturers to deploy to protect their intellectual property.
For example, in 2021, the FDA approved a generic of Otezla, a popular drug for psoriatic arthritis, but it will not hit the market until 2028. Its entry would require drugmakers to pay rebates to Medicare if they charged the program more than other developed countries for “single source” drugs and biologics. That would essentially allow the Medicare program to piggyback on other countries that negotiate the prices of some of the most expensive medicines. Those programs are still going through the rulemaking process and, again, would benefit only those covered by the Medicare program and only indirectly.
The average patient-consumer, if willing to pay cash, may find some bargains. But getting the best deal could take a lot of mixing and matching, forcing patients to become choosy shoppers, eyeing deals for essential medicines as they would for a carton of milk or eggs.
Data reporter Maia Rosenfeld contributed to this article.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF — the independent source for health policy research, polling, and journalism.












Leave a Reply