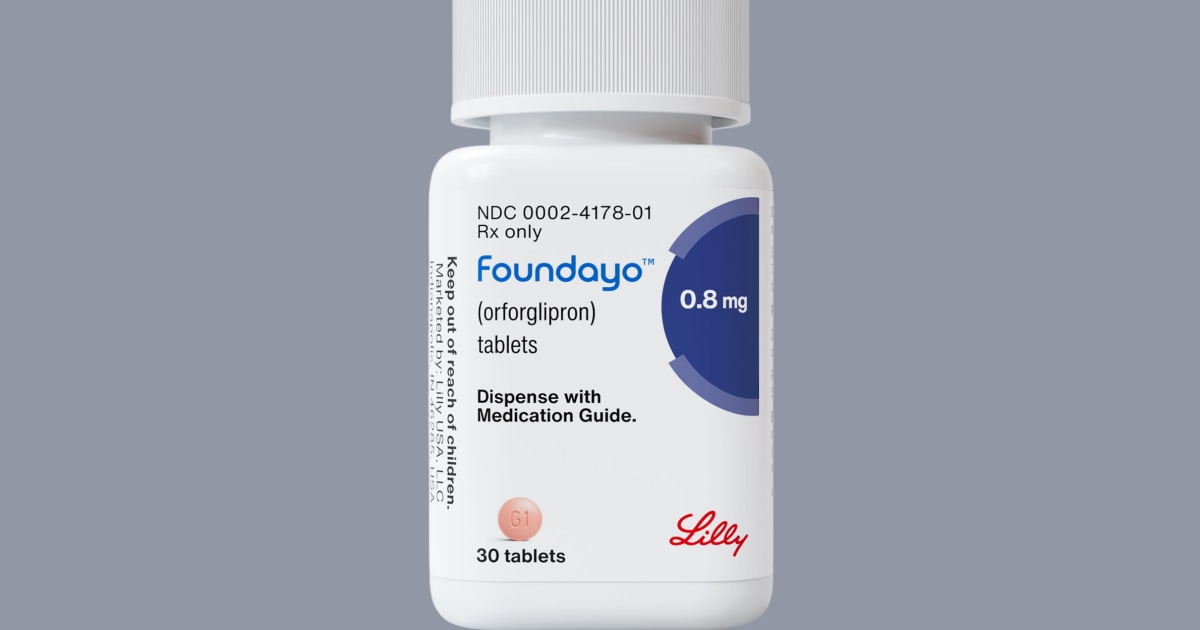
The Food and Drug Administration is asking Eli Lilly to gather more safety data on its new weight loss pill, Foundayo, including whether it could be linked to liver and heart problems, according to the approval letter published online Tuesday.
Subscribe to read this story ad-free
Get unlimited access to ad-free articles and exclusive content.
Foundayo won FDA approval this month under the Commissioner’s National Priority Voucher, a pilot program intended to fast-track drug reviews.
The pill is the second oral GLP-1 to reach the market in recent months, after Novo Nordisk’s Wegovy pill was approved in December.
In the letter, the FDA said the data it has so far isn’t enough to fully understand several potential risks tied to the drug, including heart problems such as heart attack and stroke, as well as drug-induced liver injuries. The agency also wants more data on delayed stomach emptying, which is when food remains in the stomach for too long.
Injectable GLP-1s, like Wegovy and Zepbound, carry an FDA boxed warning about a rare risk of thyroid cancer. The agency is also asking Lilly to collect data on thyroid cancer risk for at least 15 years.
In the approval letter for Novo Nordisk — the maker of the Wegovy pill — the FDA didn’t ask the company for additional data on liver or heart problems or thyroid cancer. The active ingredient in the Wegovy pill, semaglutide, however, was first approved in 2017 and has more safety data. Lilly’s drug uses a new active ingredient, called orforglipron.
Dr. Christopher McGowan, a gastroenterologist who runs a weight loss clinic in Cary, North Carolina, said the FDA often asks drugmakers to collect more safety data after approval.
“It doesn’t mean a safety problem has been found. It means they want more definitive long-term data,” McGowan said. “With orforglipron, it’s a newer, non-peptide version of a GLP-1, so we don’t yet have the same depth of safety data as the injectables.”
A spokesperson for the Department of Health and Human Services, which oversees the FDA, didn’t immediately respond to a request for comment.
A Lilly spokesperson said in an emailed statement that the FDA’s post-approval requirements are routine and consistent with the agency’s approach to ongoing safety reviews of newly approved drugs.
“Patient safety is Lilly’s top priority and we actively monitor, evaluate, and report safety information for all our medicines,” the spokesperson said.
The agency said it’s also requiring Lilly to run a series of new studies, including a registry tracking children with obesity who use weight loss drugs and a pregnancy registry to monitor outcomes.









Leave a Reply